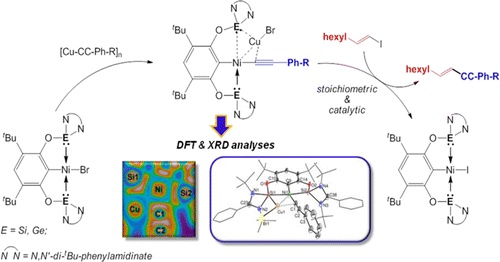
From Bis(silylene) and Bis(germylene) Pincer-Type Nickel(II) Complexes to Isolable Intermediates of the Nickel-Catalyzed Sonogashira Cross-Coupling Reaction | The Hartwig Group
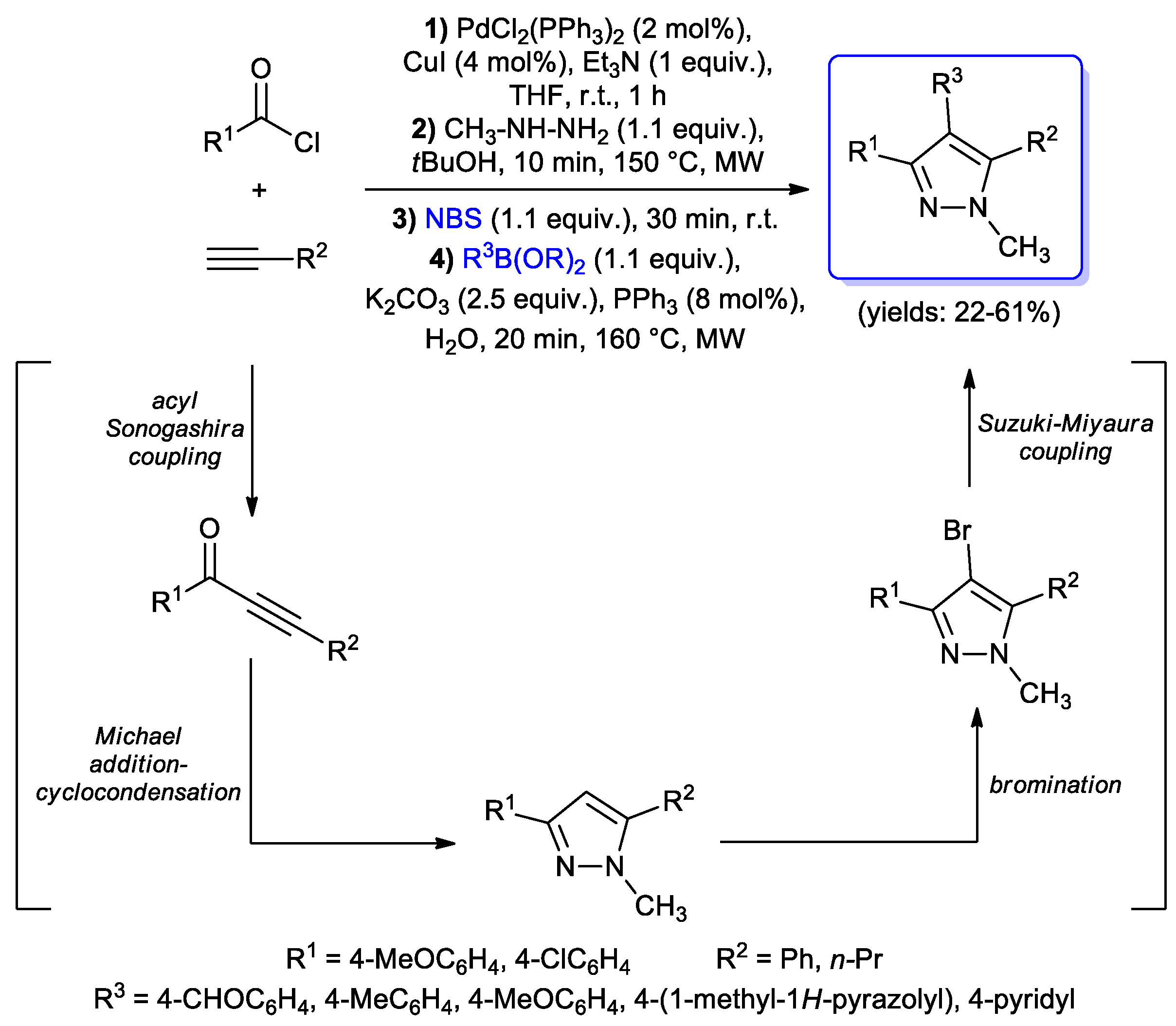
Catalysts | Free Full-Text | Acyl Sonogashira Cross-Coupling: State of the Art and Application to the Synthesis of Heterocyclic Compounds | HTML
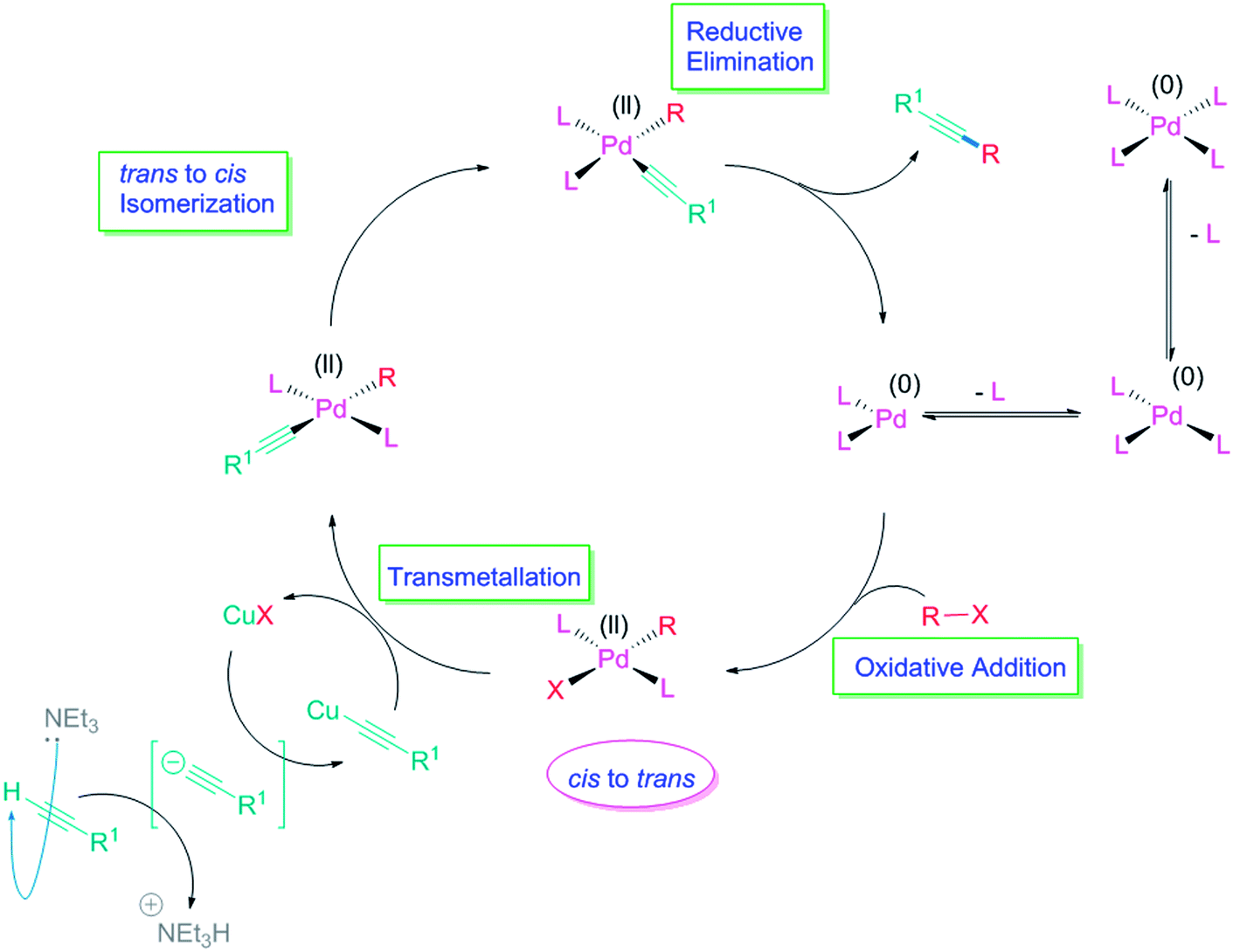
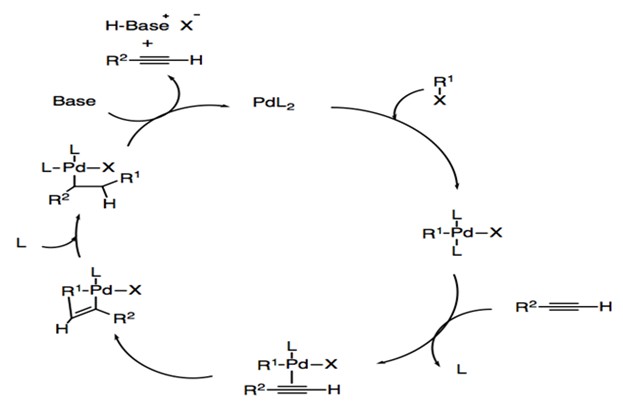
Recent mechanistic developments and next generation catalysts for the Sonogashira coupling reaction - RSC Advances (RSC Publishing) DOI:10.1039/C4RA09105A

Insights into Sonogashira Cross‐Coupling by High‐Throughput Kinetics and Descriptor Modeling - an der Heiden - 2008 - Chemistry – A European Journal - Wiley Online Library
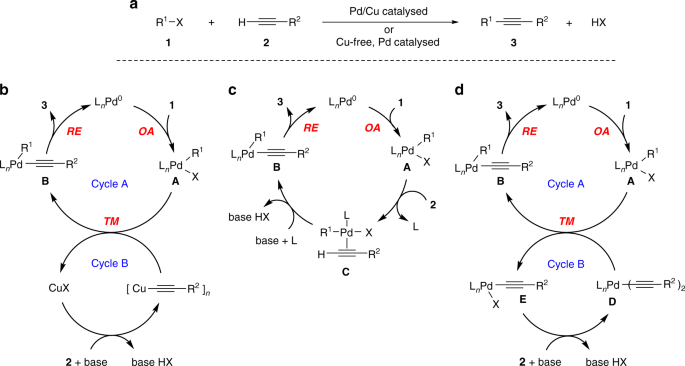
Mechanism of copper-free Sonogashira reaction operates through palladium-palladium transmetallation | Nature Communications

Mechanism of copper-free Sonogashira reaction operates through palladium-palladium transmetallation | Nature Communications

Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross- Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Copper-catalyzed enantioselective Sonogashira-type oxidative cross-coupling of unactivated C(sp3)−H bonds with alkynes | Nature Communications

The Sonogashira reaction. a General representation of Pd/Cu catalysed... | Download Scientific Diagram