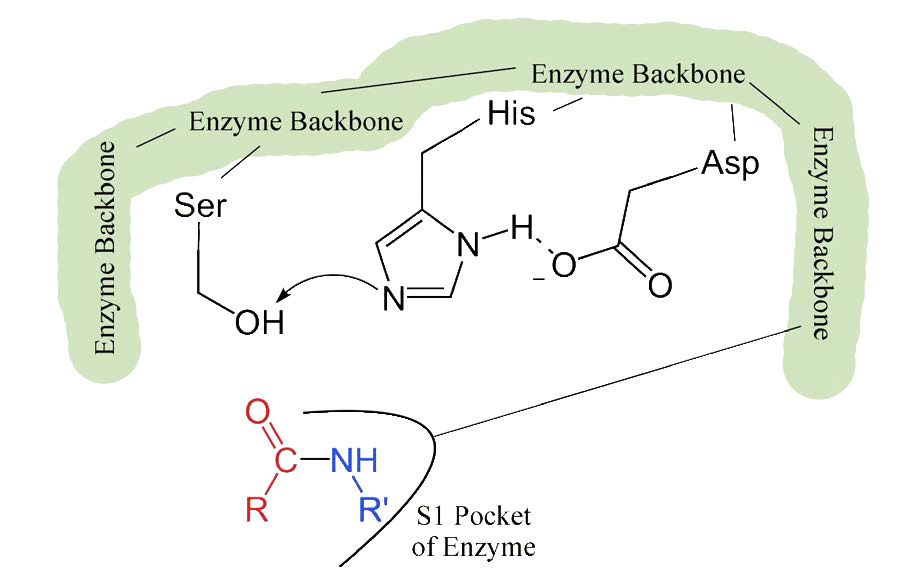
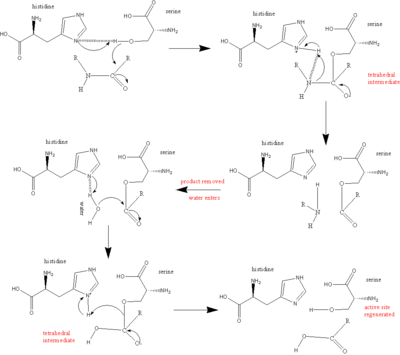
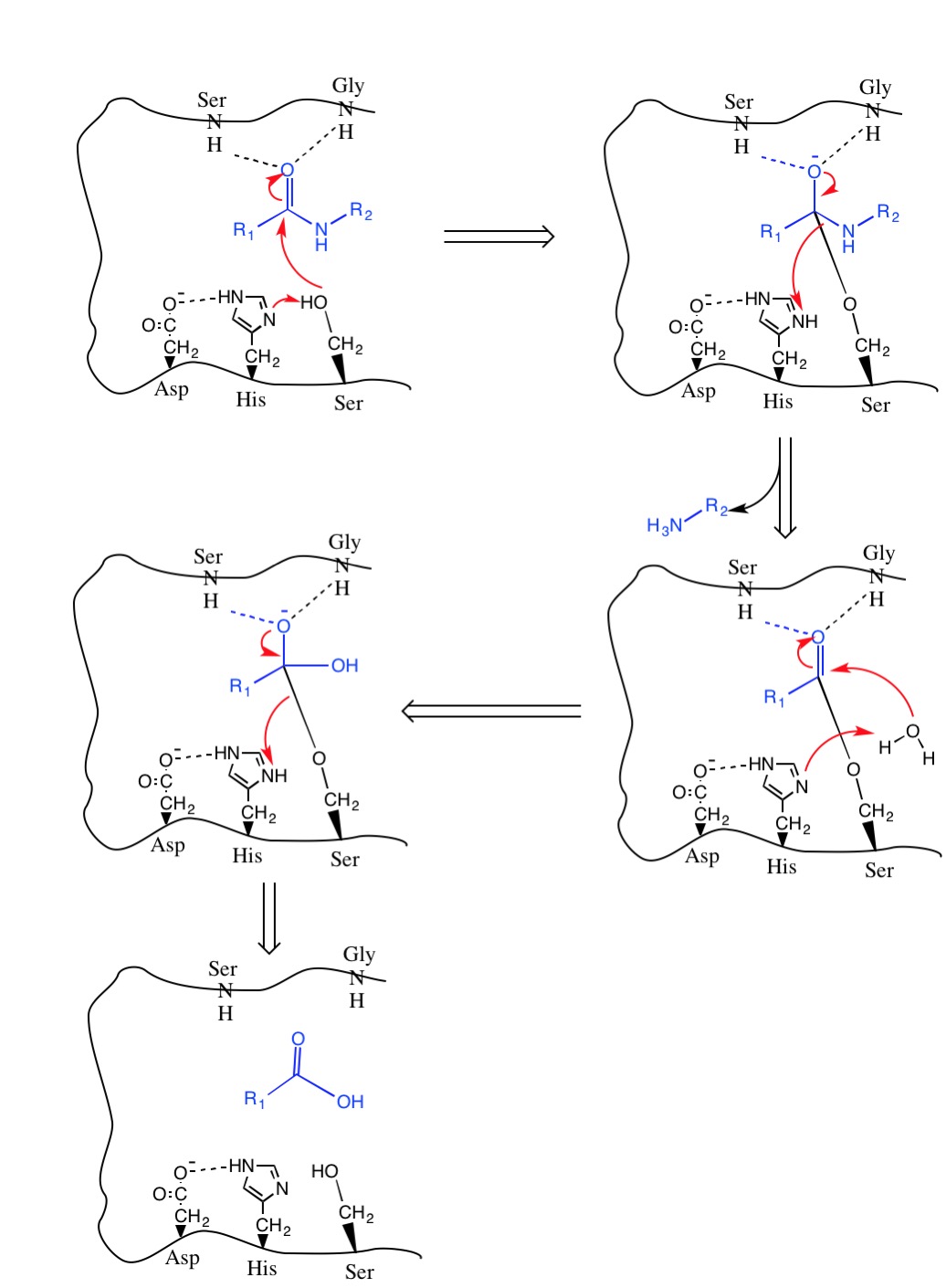
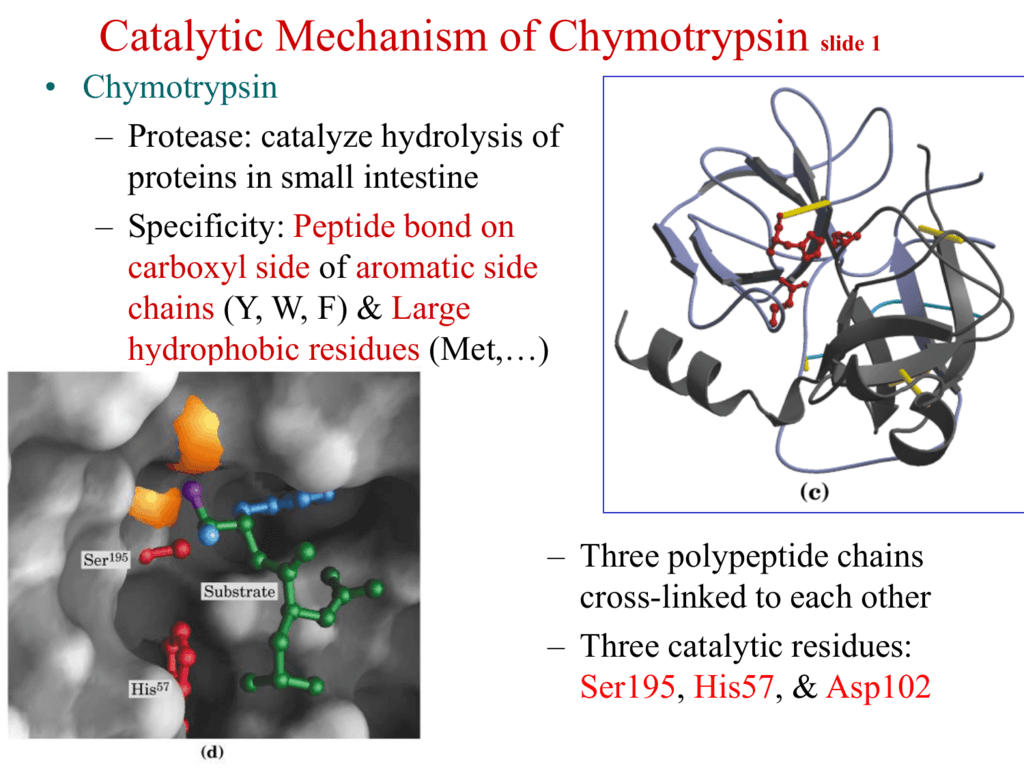
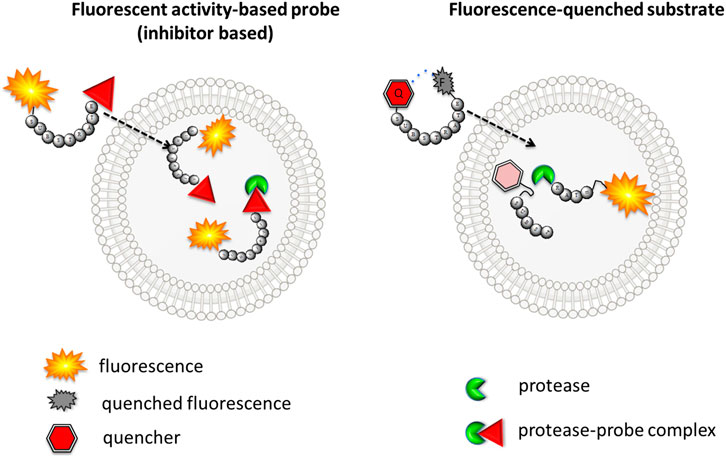
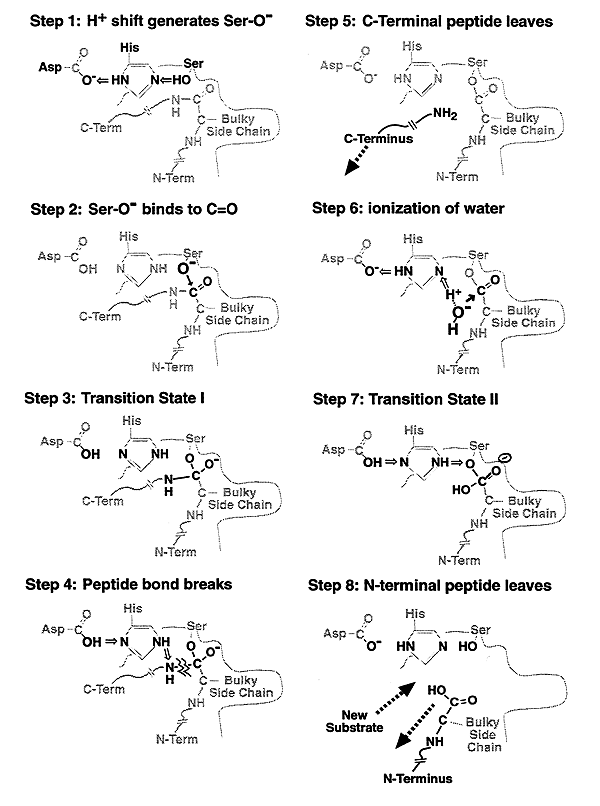
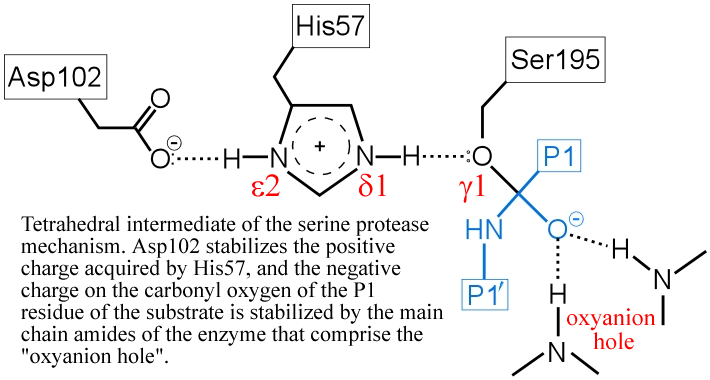
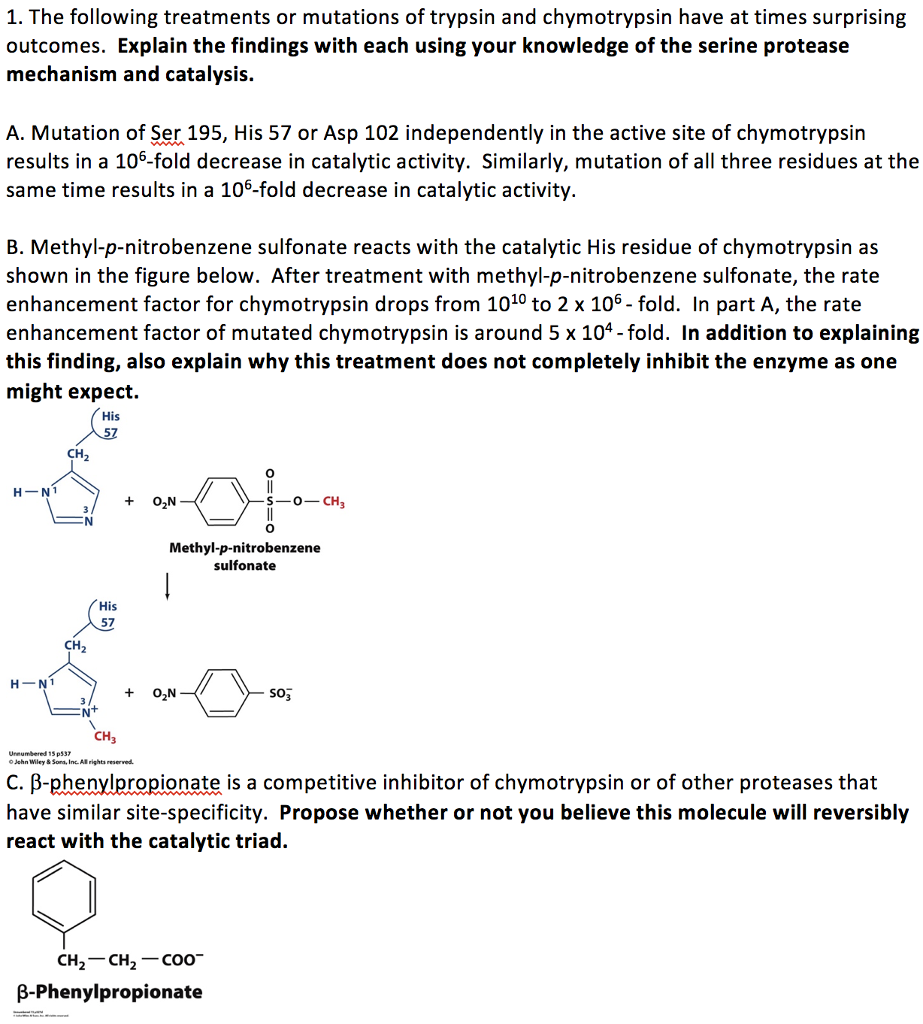
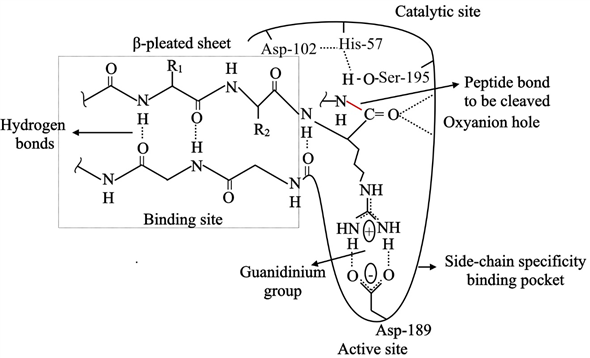
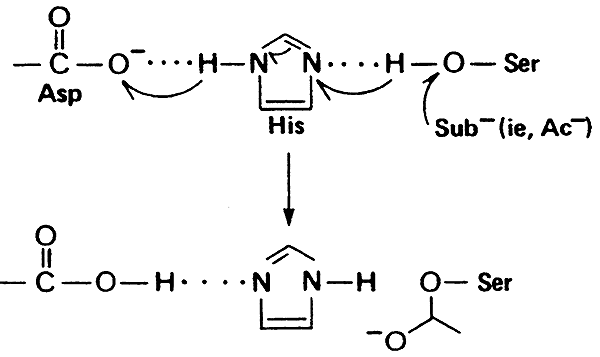
Evolutionary Divergence of Substrate Specificity within the Chymotrypsin-like Serine Protease Fold* - Journal of Biological Chemistry
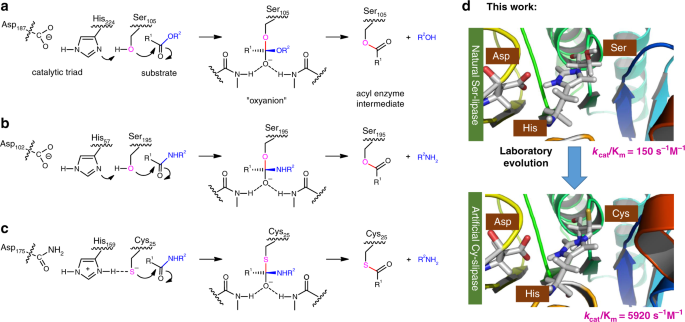
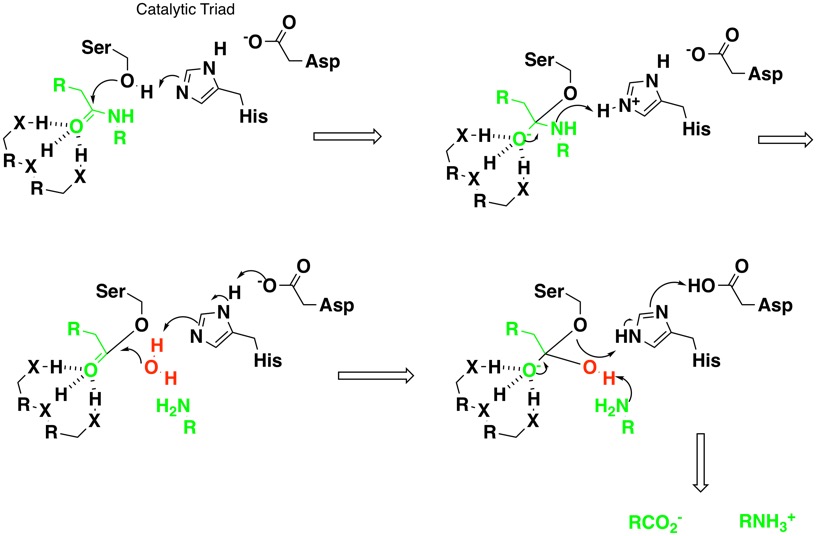
Artificial cysteine-lipases with high activity and altered catalytic mechanism created by laboratory evolution | Nature Communications

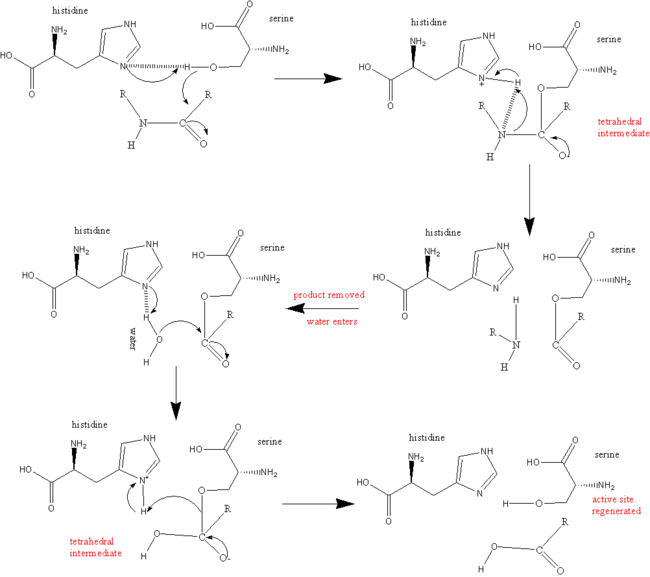
reaction mechanism of a serine protease. The catalytic triad of the... | Download Scientific Diagram