Chain length affects pancreatic lipase activity and the extent and pH–time profile of triglyceride lipolysis - ScienceDirect
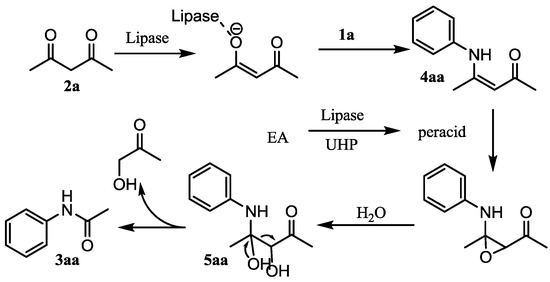
Catalysts | Free Full-Text | Lipase-Mediated Amidation of Anilines with 1,3-Diketones via C–C Bond Cleavage | HTML
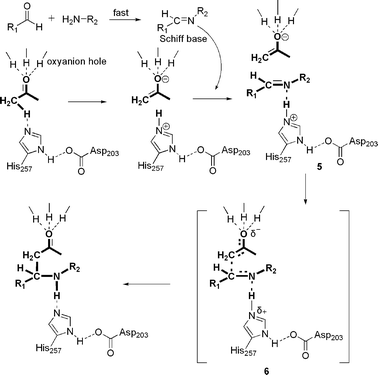
Lipase-catalysed direct Mannich reaction in water : utilization of biocatalytic promiscuity for C–C bond formation in a “one-pot” synthesis - Green Chemistry (RSC Publishing) DOI:10.1039/B817524A

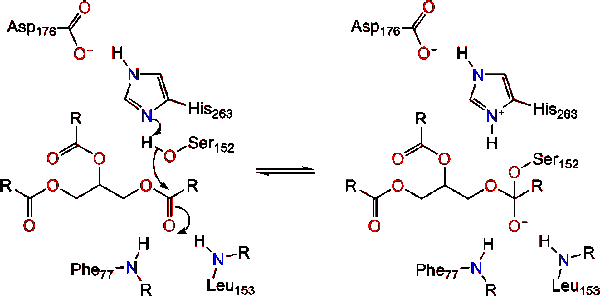
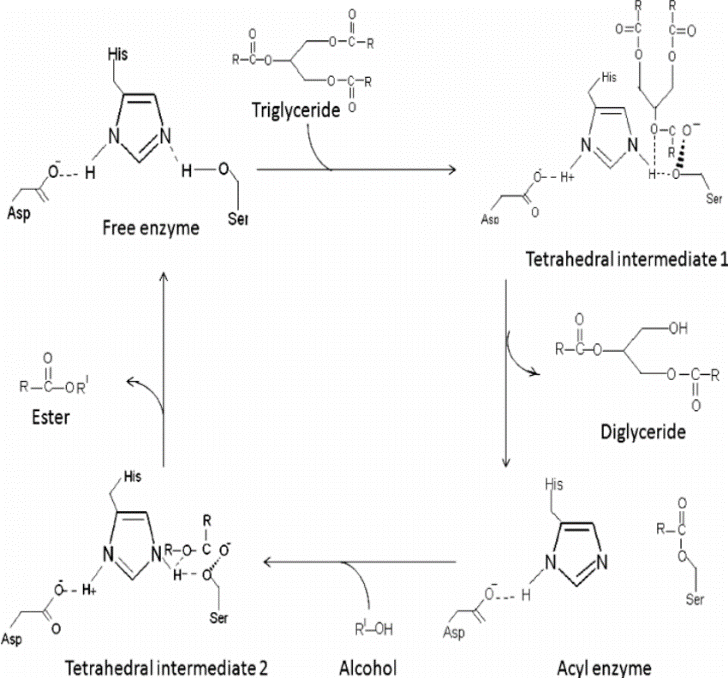
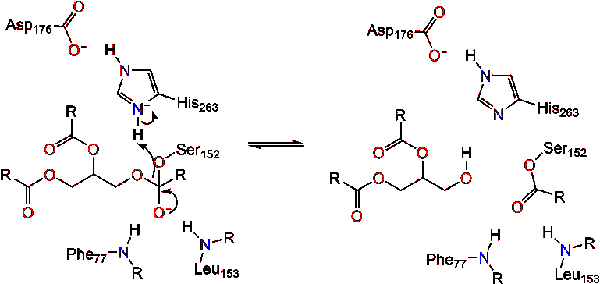
Mechanism of lipase catalysis. The active site of the enzyme contains a... | Download Scientific Diagram
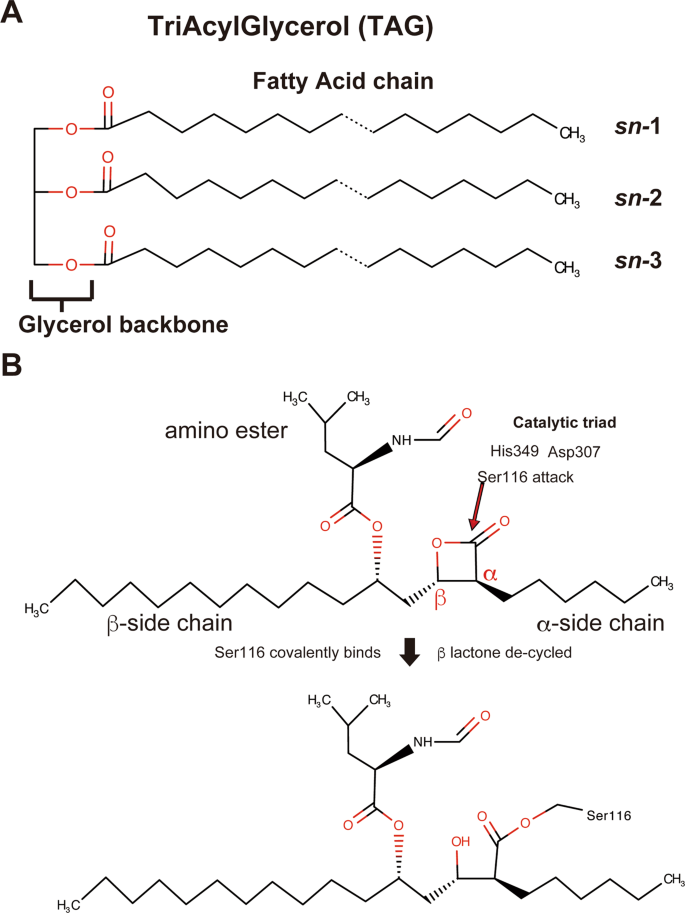
Crystal structure of pathogenic Staphylococcus aureus lipase complex with the anti-obesity drug orlistat | Scientific Reports
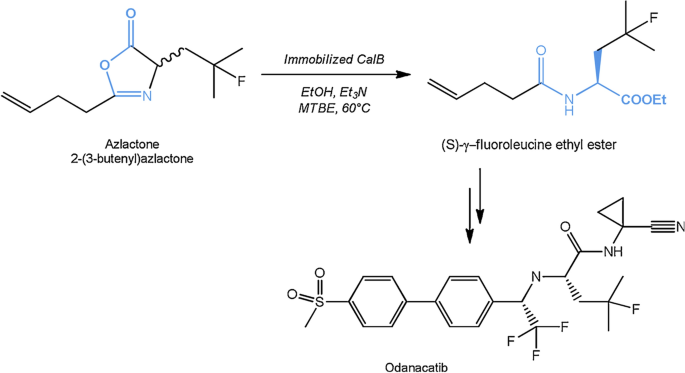
Microbial lipases and their industrial applications: a comprehensive review | Microbial Cell Factories | Full Text
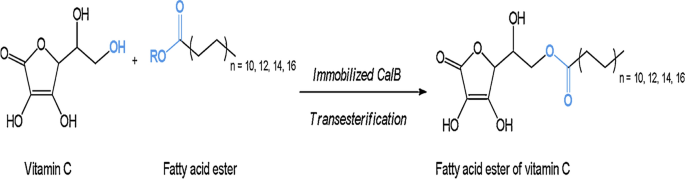
Microbial lipases and their industrial applications: a comprehensive review | Microbial Cell Factories | Full Text

Discovery of novel amide tripeptides as pancreatic lipase inhibitors by virtual screening - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C8NJ05884A
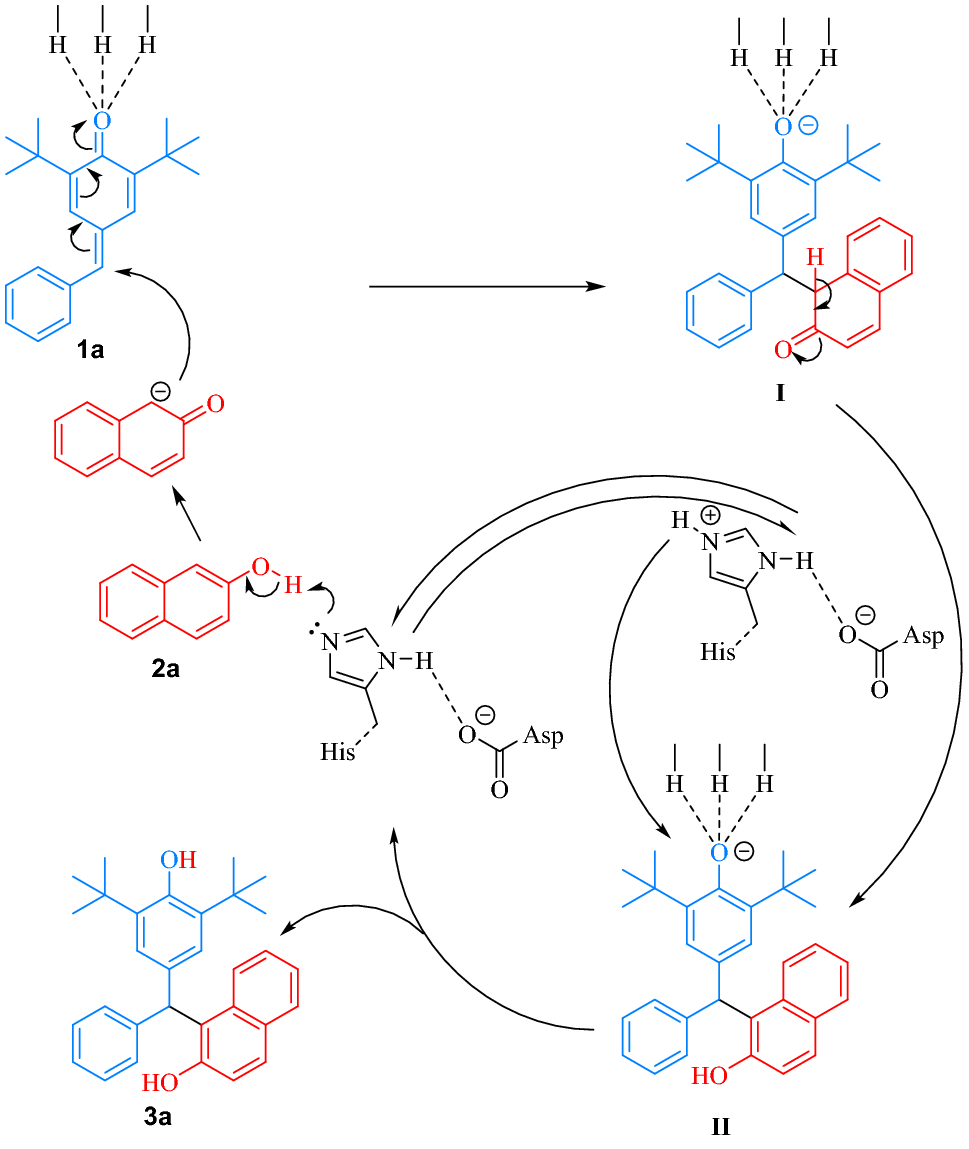
Figure 5 | Lipase-Catalyzed Highly Efficient 1,6-Conjugated Addition for Synthesis of Triarylmethanes | SpringerLink
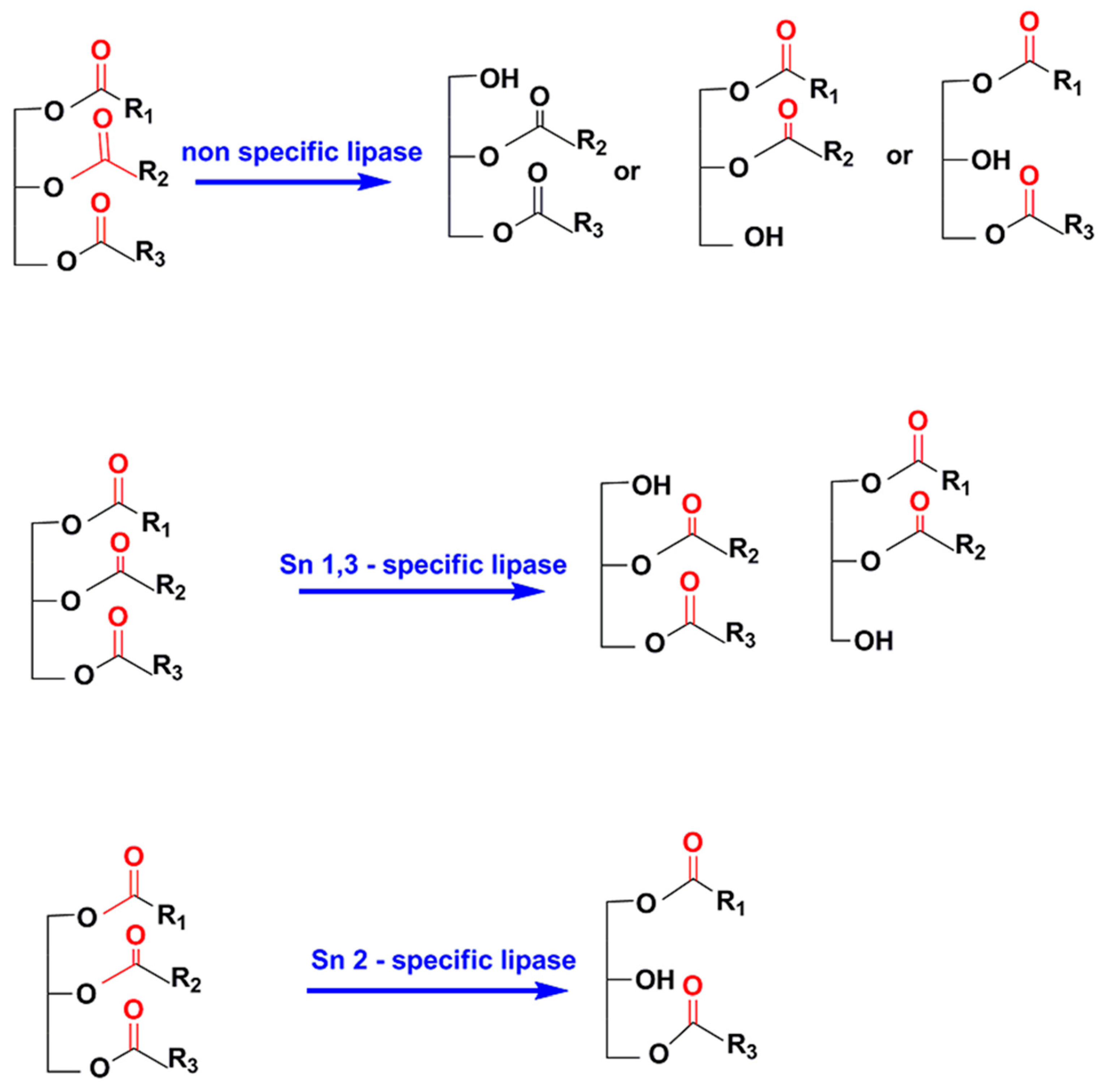
Catalysts | Free Full-Text | Main Structural Targets for Engineering Lipase Substrate Specificity | HTML


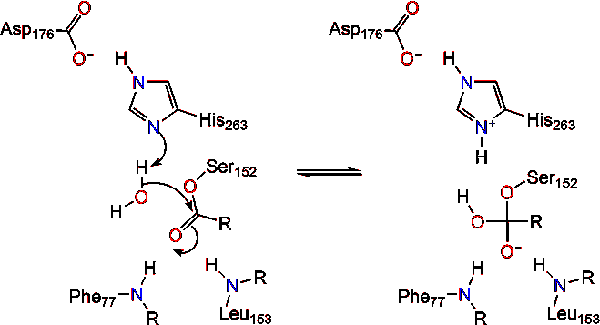



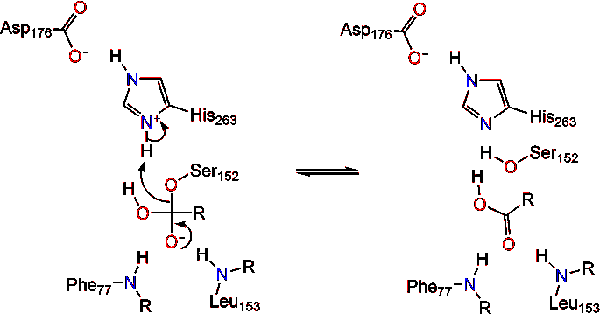



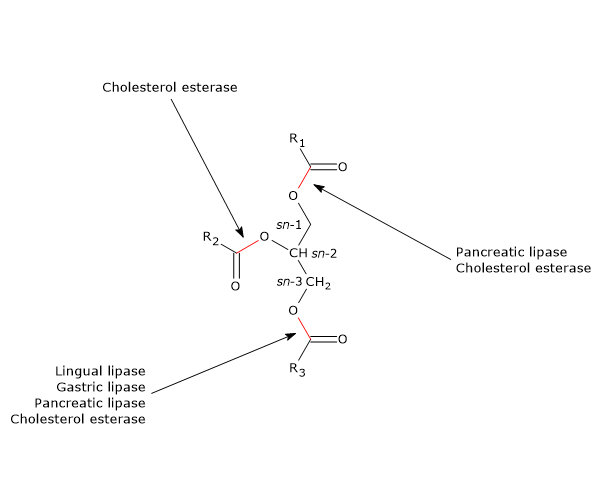

.png)