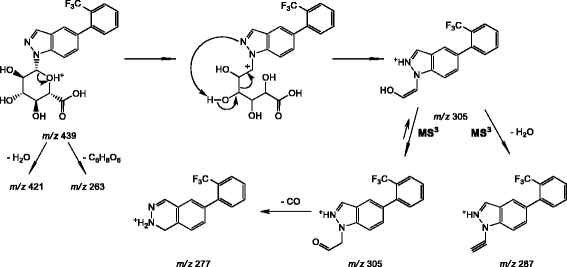
Human UDP-glucuronosyltransferase UGT1A4 forms tertiary N-glucuronides predominately with the energetically less favored tautomer of substituted 1H-indazole (benzpyrazole) | Journal of Analytical Science and Technology | Full Text
![PDF] Glucuronidation as a mechanism of intrinsic drug resistance in colon cancer cells: contribution of drug transport proteins. | Semantic Scholar PDF] Glucuronidation as a mechanism of intrinsic drug resistance in colon cancer cells: contribution of drug transport proteins. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6e80f1975291df8420e6c40ff72a66972225ffec/2-Figure1-1.png)
PDF] Glucuronidation as a mechanism of intrinsic drug resistance in colon cancer cells: contribution of drug transport proteins. | Semantic Scholar
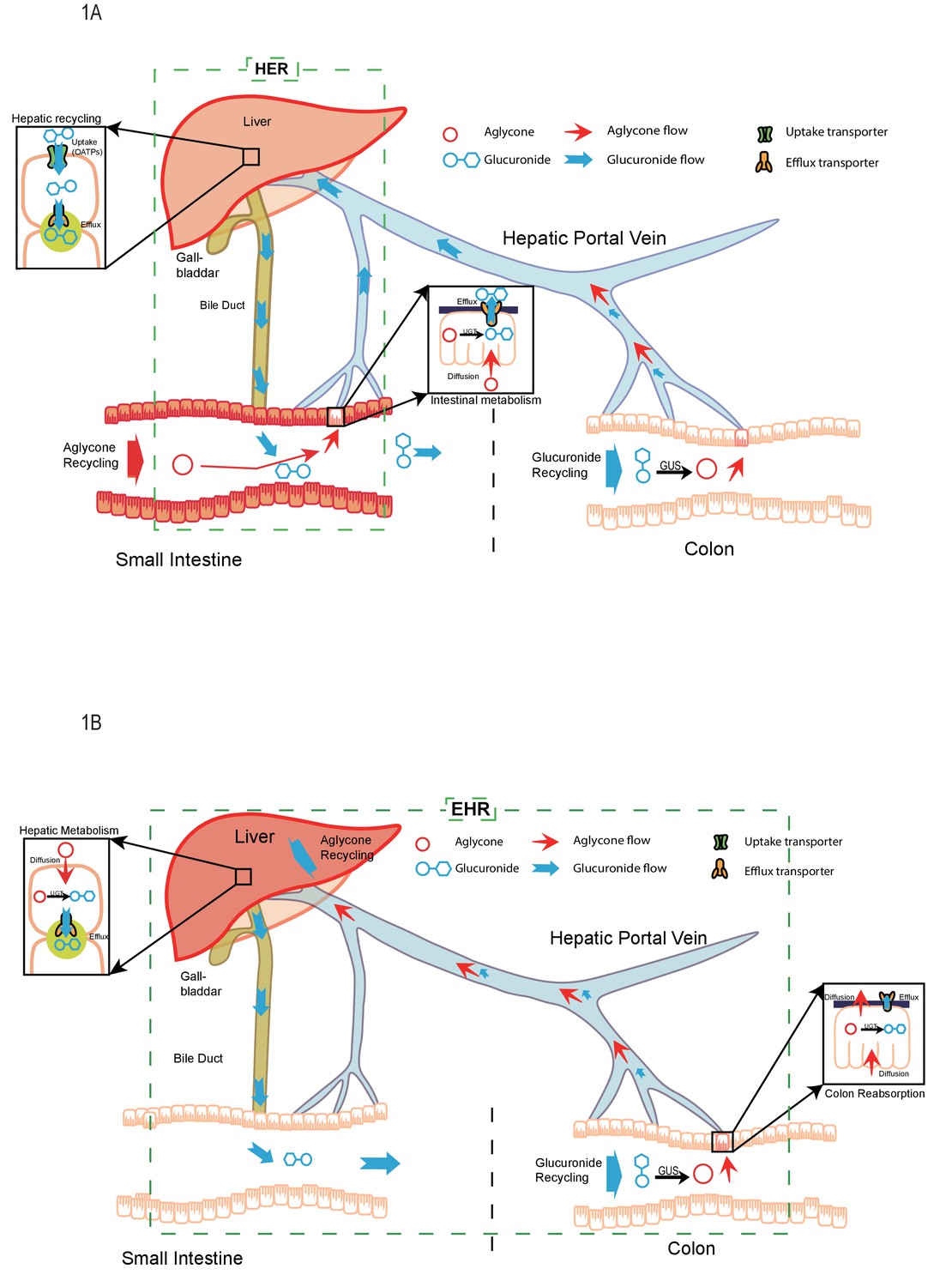
Hepatoenteric recycling is a new disposition mechanism for orally administered phenolic drugs and phytochemicals in rats | eLife

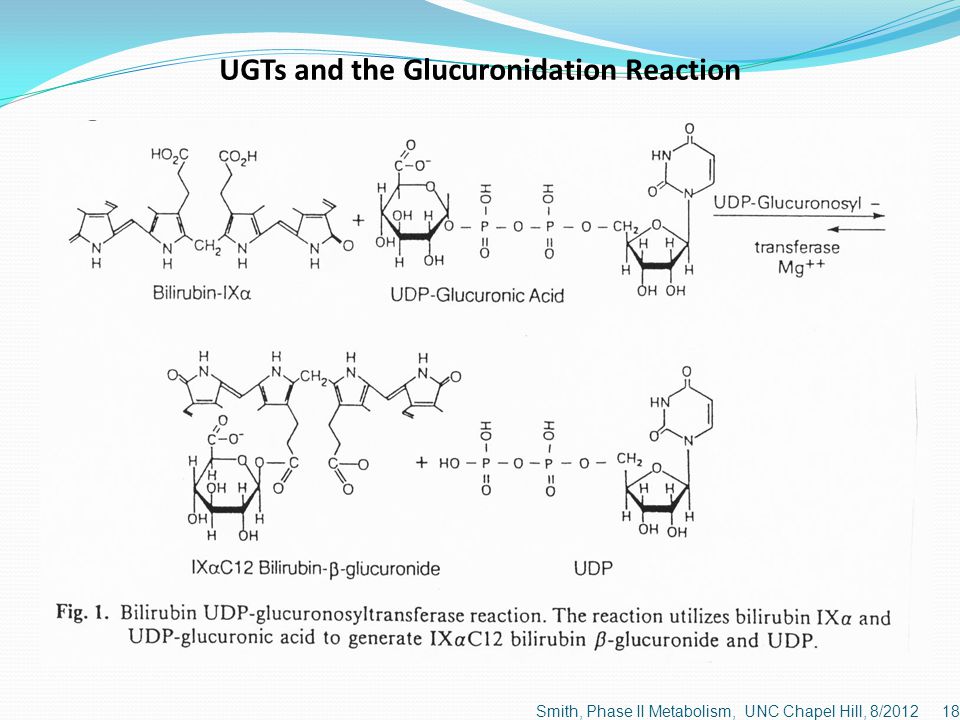
Challenges and Opportunities with Predicting In Vivo Phase II Metabolism via Glucuronidation From In Vitro Data | SpringerLink

Diagram showing glucuronidation of phenols by nucleophilic at- tacking... | Download Scientific Diagram

Figure 1 from 2D QSAR Study for Gemfibrozil Glucuronide as the Mechanism-based Inhibitor of CYP2C8 | Semantic Scholar

Methylation, Glucuronidation, and Sulfonation of Daphnetin in Human Hepatic Preparations In Vitro: Metabolic Profiling, Pathway Comparison, and Bioactivity Analysis - Journal of Pharmaceutical Sciences

Sulfinpyrazone C-Glucuronidation Is Catalyzed Selectively by Human UDP-Glucuronosyltransferase 1A9 | Drug Metabolism & Disposition

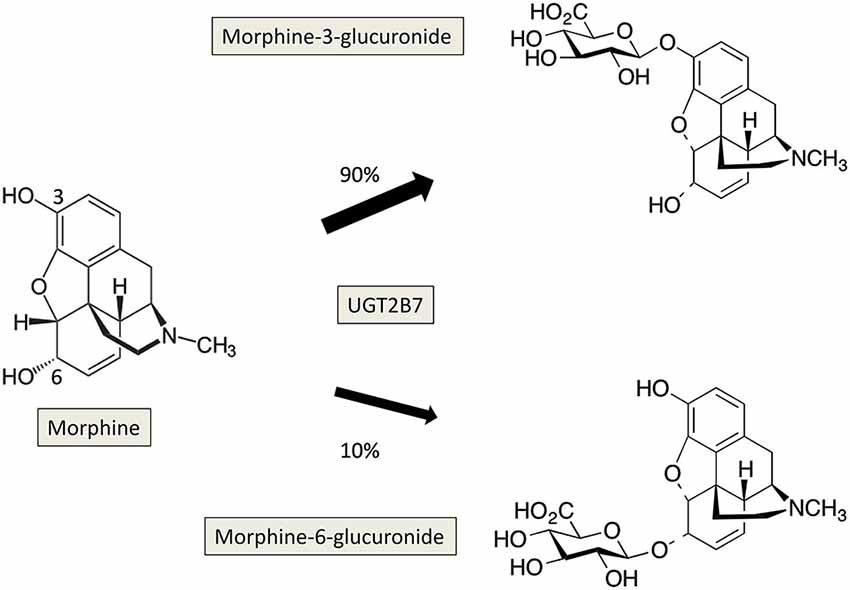
The regioselective glucuronidation of morphine by dimerized human UGT2B7, 1A1, 1A9 and their allelic variants | Acta Pharmacologica Sinica
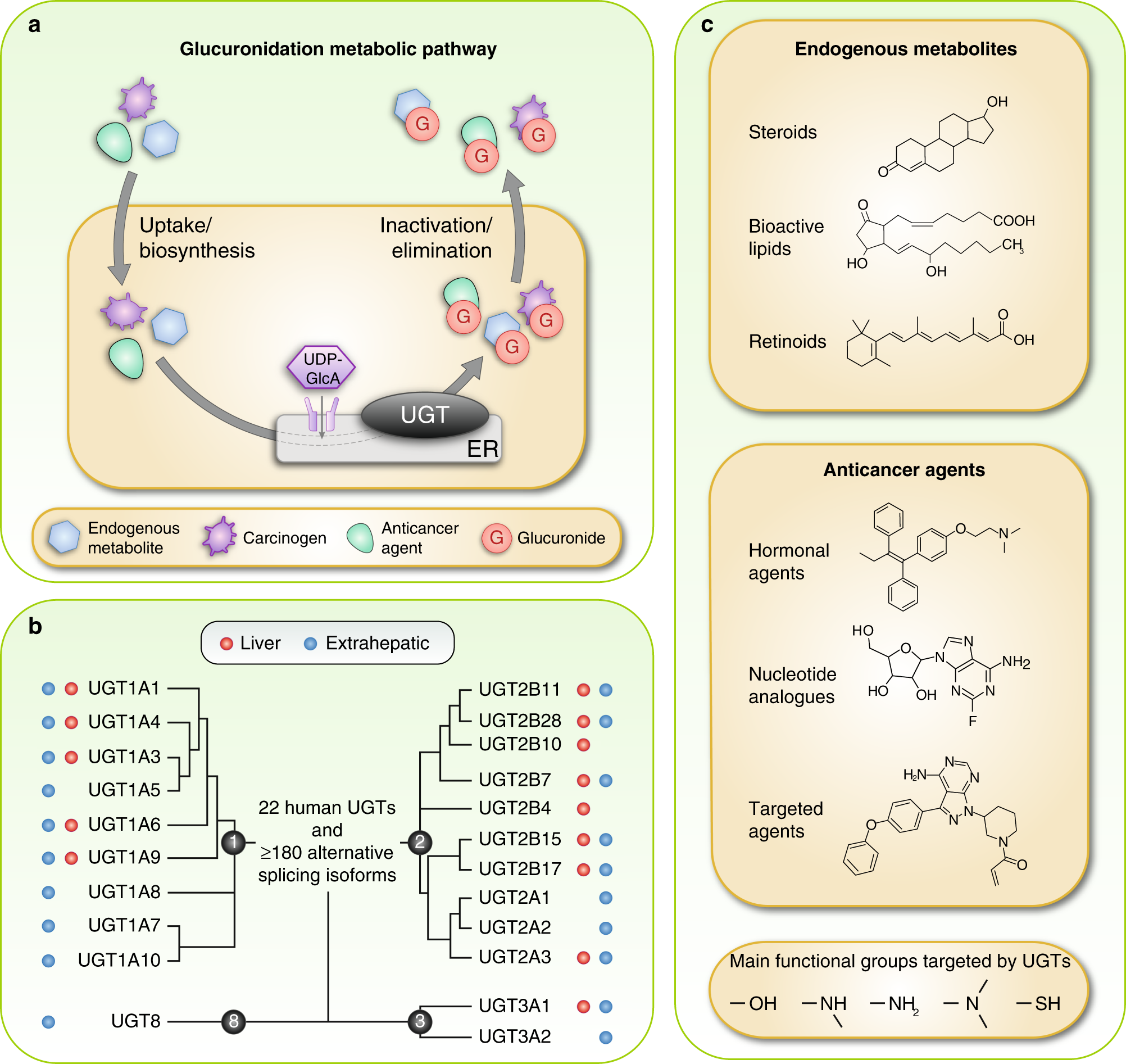
Emerging roles for UDP-glucuronosyltransferases in drug resistance and cancer progression | British Journal of Cancer

Identification of Human UGT2B7 as the Major Isoform Involved in the O- Glucuronidation of Chloramphenicol | Drug Metabolism & Disposition
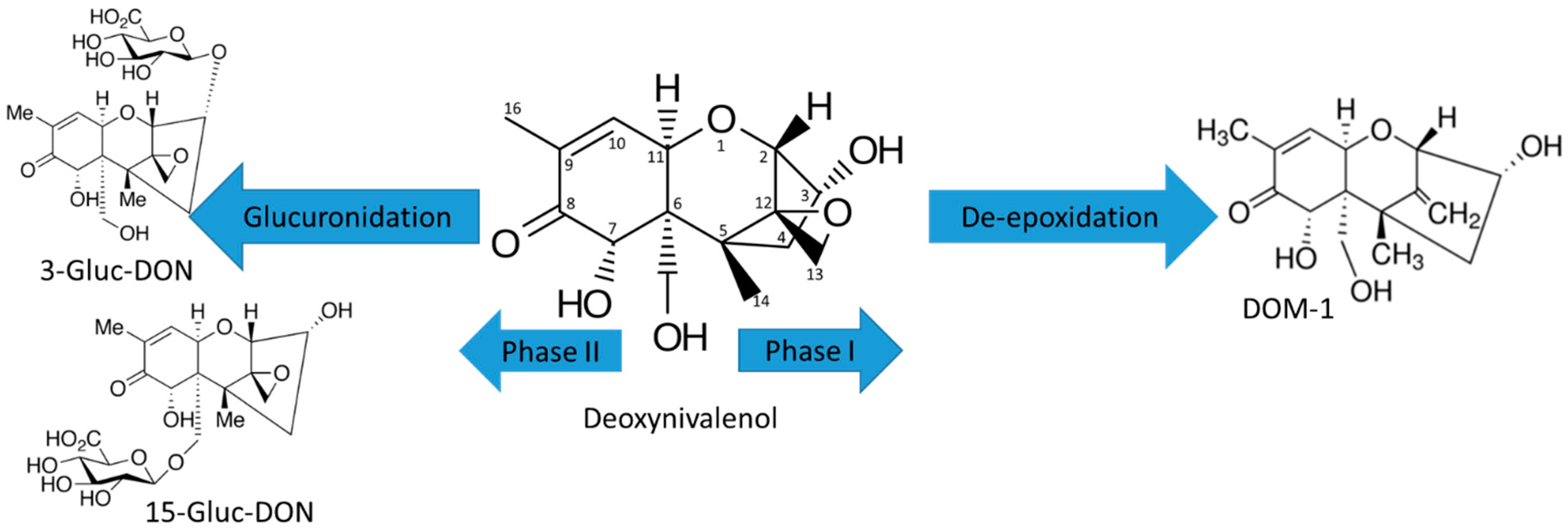
Toxins | Free Full-Text | Characterization of Phase I and Glucuronide Phase II Metabolites of 17 Mycotoxins Using Liquid Chromatography—High-Resolution Mass Spectrometry | HTML