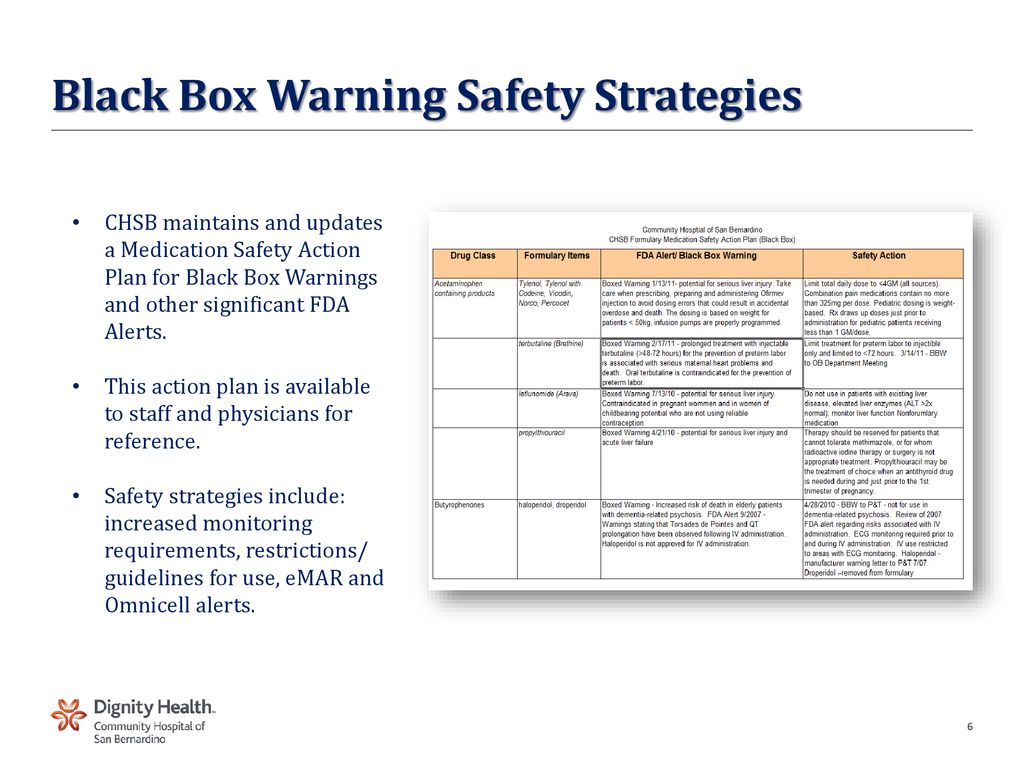
Cymbalta Dangers International - FDA Black Box Warning Label Definition and Safety Awareness (Rx Drugs 101). "The FDA Black Box” warning label (sometimes called “black label” or “boxed” warning) is a type

Black Box Warning Contraindicated Comedications: Concordance Among Three Major Drug Interaction Screening Programs | Semantic Scholar